|
(Of note, neonatal ICU and nursery locations require stratification by birth weight, and specialty areas such as dialysis and oncology require stratification by temporary and permanent central lines). Each patient only counts once, even if they have more than one central line. To collect these denominator days (regardless of whether using rates or SIRs), each unit provides a count at the same time every day of how many patients on the unit have one or more central lines at that specific point in time. More recently, public reporting has created a need for a standardized infection ratio (SIR) which allows for a single number to be used to reflect overall hospital CLABSI performance while allowing appropriate risk adjustment to take place to account for differing patient populations and other facility considerations. Each unit has a mean rate of infection to which comparisons are made. Central line days are used as the standardized denominator for reporting CLABSI across units, expressed as the number of CLABSI/1000 central line days historically. A pathogen requires only one positive culture to be considered an infection organisms identified as commensals (i.e., coagulase-negative staphylococcus or diphtheroids) require two positive cultures collected at different times, in addition to clinical symptoms of infection such as fever or hypotension.Įqually important to the above infection surveillance is the collection of accurate denominators to allow for risk-adjusted CLABSI rates and ratios. There are specific criteria to be followed when determining whether a suspected infection can be labeled as a secondary infection rather than a CLABSI. Pathogens do not require symptoms in addition to the positive culture, but the patient record must be reviewed to rule out other potential sources of infection, which may cause the bacteremia to be defined as secondary to another site of infection. Based on whether the organism is classified as a pathogen or a commensal (defined by CDC), the appropriate next steps are followed. 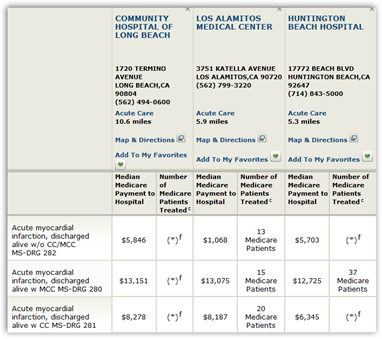
The most common starting point is through review of positive blood cultures. Surveillance is an active process which requires proactively reviewing data sources indicative of an infection rather than relying on coded data or provider self-reporting of infections. 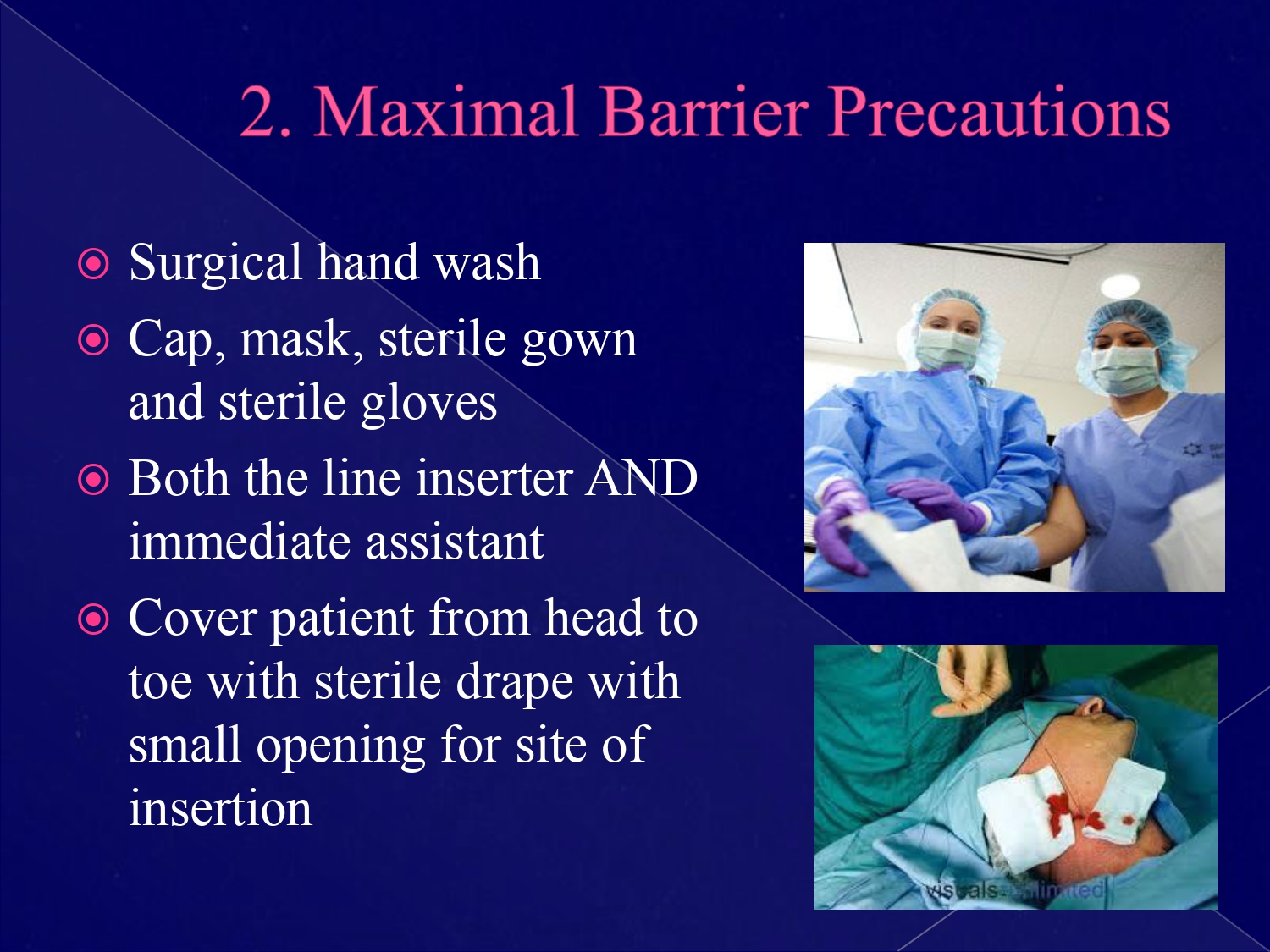
Surveillance for CLABSIs is recommended in all areas of healthcare facilities that care for patients with CVADs and is required by Joint Commission to be conducted facility-wide rather than in a targeted manner (T. With minor variations, the Australian Commission on Safety and Quality in Health Care has implemented the same definitions throughout their country (Healthcare 2015). They are also used for reporting into Press Ganey/National Database of Nursing Quality Indicators (NDNQI), which serves as the data repository for hospitals seeking Magnet™ designation. These definitions are reviewed and may be slightly revised annually and serve as the standard surveillance criteria for infections included in public reporting mandates through state regulations and federal Centers for Medicare and Medicaid Services (CMS) requirements for value-based purchasing (CDC 2017). The National Healthcare Safety Network (NHSN) of the Centers for Disease Control and Prevention (CDC) publishes surveillance definitions for a broad range of healthcare-associated infections (HAI) (Horan et al. 
This chapter aims to define how surveillance is performed and the various uses of the gathered/reported information. The surveillance data is used in a variety of ways including rating of hospitals for patient satisfaction, reimbursement/value-based purchasing percentages, comparison to other hospitals of similar bed size, central line device utilization ratio to determine if a hospital is using an inordinate ratio of central catheters, and determination of a standardized infection ratio. 
Other parts of the world do not have mandatory reporting. Based on the model of surveillance in the United States, if it is determined the bloodstream infection is associated with the central venous access device, mandatory data is collected. Surveillance is an active process which requires proactively reviewing data sources indicative of an infection with the most common starting point through review of positive blood cultures.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
